Certificates and Compliance
Demonstrating Quality, Safety, and Global Regulatory Alignment
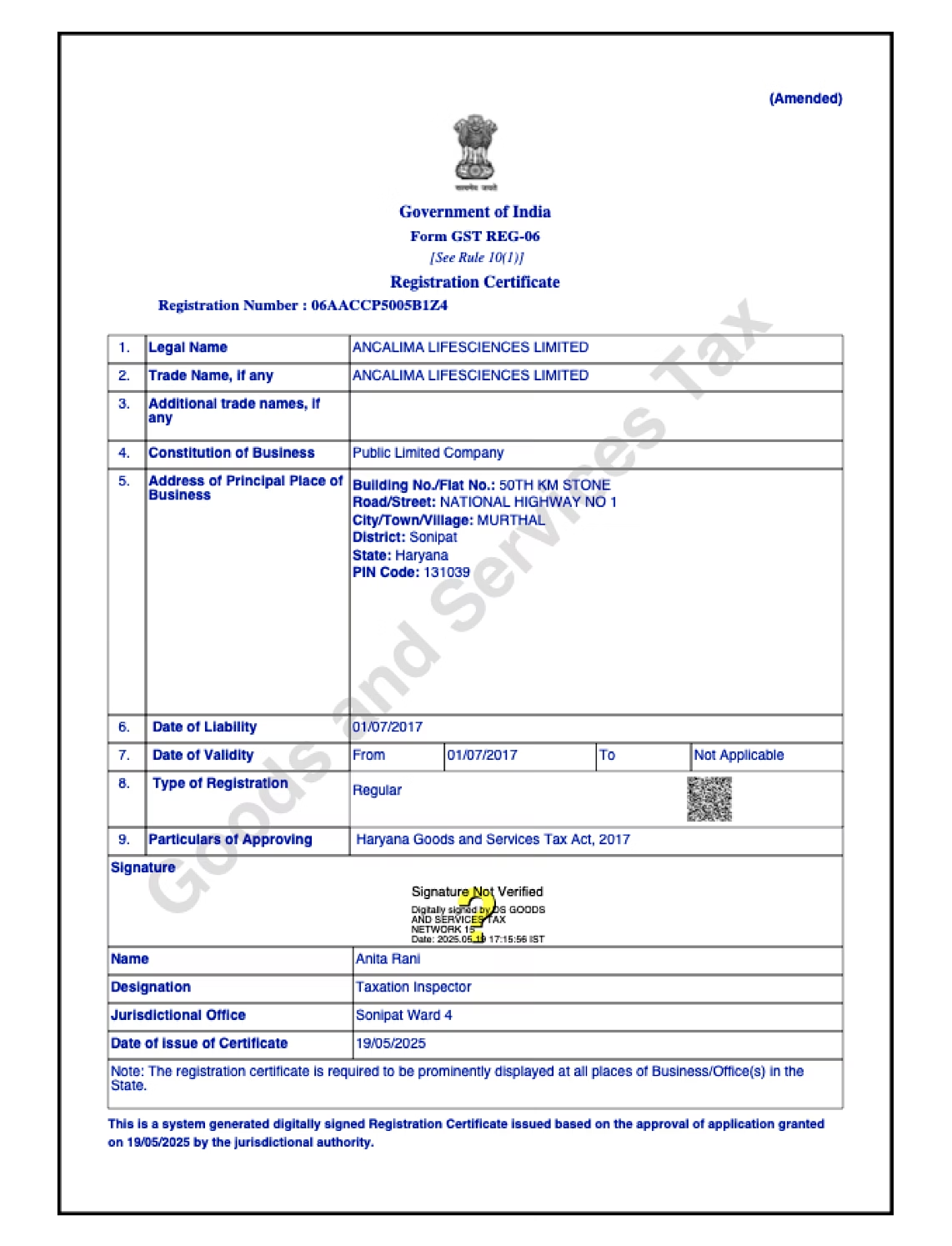
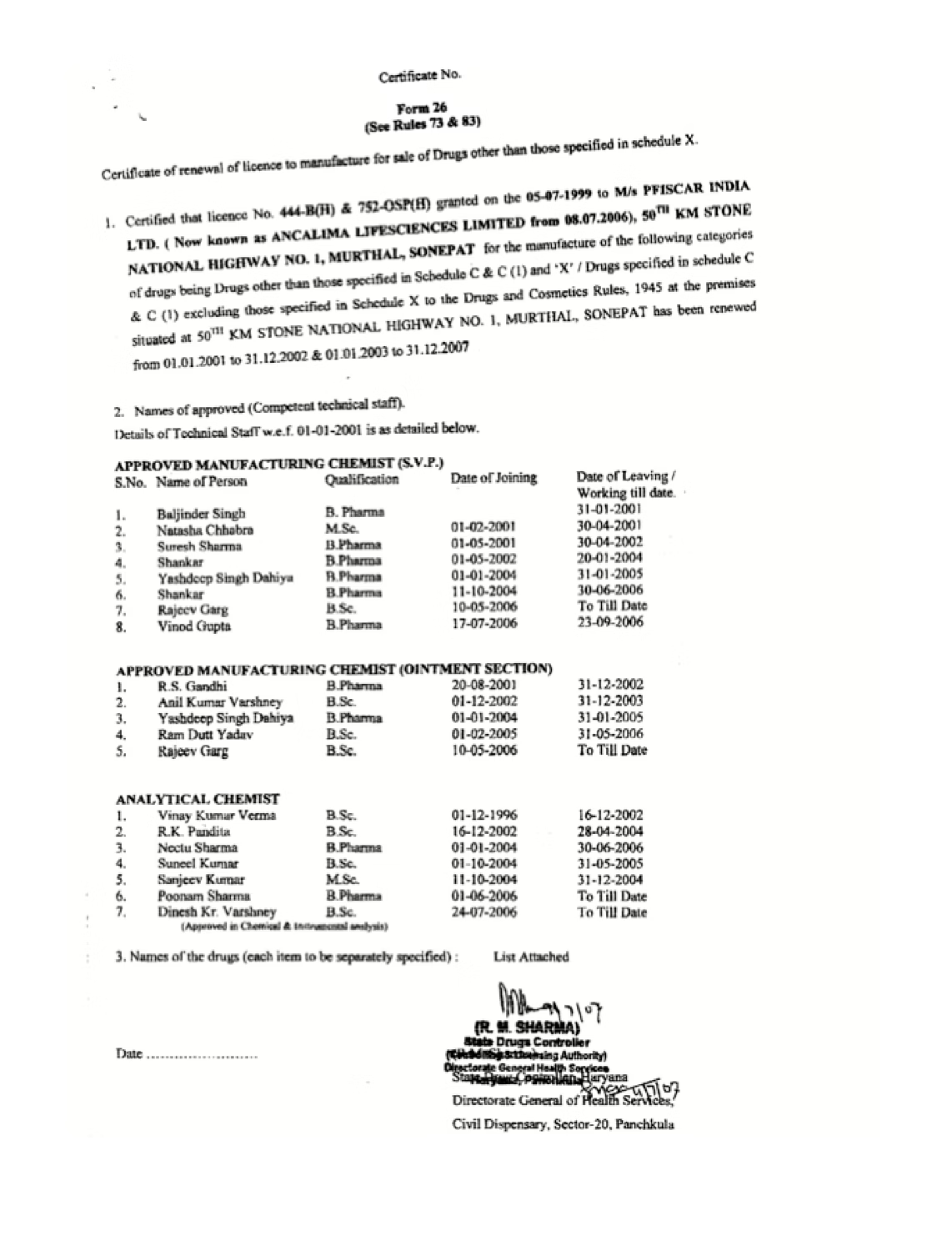
At Ancalima Lifesciences, certifications and compliance are fundamental to how we operate and how we build trust with global partners. Our manufacturing and export activities are governed by recognized international standards, ensuring that products consistently meet regulatory, safety, and quality expectations across global markets.
We view compliance not as a requirement alone, but as a commitment to excellence, transparency, and accountability.
Internationally Recognized Certifications
Ancalima Lifesciences maintains certifications that reflect adherence to globally accepted manufacturing and quality standards. These certifications validate our processes, facilities, and systems, providing confidence to international buyers, regulators, and partners.
Our certified frameworks support pharmaceutical and cosmetic manufacturing, quality assurance, and export readiness, enabling us to serve diverse markets efficiently and responsibly.


Regulatory Compliance Framework
Compliance at Ancalima Lifesciences is supported by structured systems, documented procedures, and continuous oversight. All operations are conducted in alignment with applicable regulatory requirements and industry guidelines.
We ensure that manufacturing, testing, documentation, and release activities are consistently reviewed and maintained in accordance with current regulatory expectations.
Audit Readiness & Documentation Control
Audit readiness is an integral part of our compliance culture. Ancalima maintains controlled documentation systems that support traceability, data integrity, and regulatory review.
Our teams are trained to respond effectively to audits, inspections, and customer assessments, ensuring transparency and reliability at all times.


Continuous Compliance & Improvement
Regulatory environments evolve, and Ancalima Lifesciences remains proactive in monitoring changes, updating systems, and enhancing processes to maintain ongoing compliance and operational excellence.
Our Certificate